ACE2 (Guide to Pharmacology Target id: 1614) normally functions as an enzyme metabolising peptides that regulate the cardiovascular system. As is well known though, it has recently gained additional scientific fame by also acting as a receptor for SARS-CoV-2. This study [1] compared the distribution in human tissue of ACE2 versus deltaACE2, a recently discovered truncated isoform that has lost the spike binding sites for SARS-CoV-2, and is predicted to confer reduced susceptibility to viral infection. ACE2 was highly expressed in human lung, kidney, heart, liver and vasculature. However, deltaACE2 was the predominant protein alternative initiation variant in lung airway and liver bile duct epithelia, which may be a contributing factor to variation in response to the virus. A diagrammatic relationships of the full length and dACE is shown below.
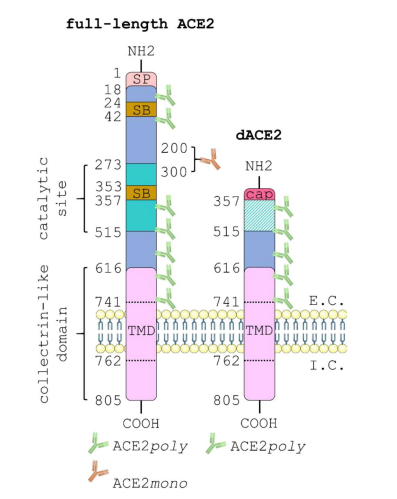
The discovery of this novel short dACE2 variant in two independent reports may have important implications in COVID-19 research. It is enzymatically inactive, and the absence of the spike binding domains of the full-length ACE2 suggests dACE2 does not bind SARS-CoV-2 spike protein. It is still unclear what the precise effects of such an alternative form are in both normal physiology and in relation to SARS-CoV-2 infection and Long COVID, but the ratio of dACE2 versus the full-length protein may be a contributing factor in the wide inter-individual variation in response to COVID-19
- Williams TL, Strachan G, Macrae RGC, Kuc RE, Nyimanu D, Paterson AL, Sinha S, Maguire JJ, and Davenport AP. Differential expression in humans of the viral entry receptor ACE2 compared with the short deltaACE2 isoform lacking SARS-CoV-2 binding sites. Sci Rep. 11:24336. (2021). [PMID: 34934117]
Contributed by Christopher Southan, ORCID 0000-0001-9580-0446, Data Science, Medicines Discovery Catapult and Honorary Fellow, University of Edinburgh


Leave a comment