The GtoPdb team need no convincing about the importance of eliminating equivocality in gene and gene product names, as they are used in the pharmacology, chemical biology and drug discovery literature. Crucially, this also applies to their inclusion in curated database bioactivity records such as GtoPdb. We were thus particularly pleased to see “Standardizing gene product nomenclature” that appeared recently in PNAS [1]. This included not only an honourable mention of the Nomenclature Committee of the International Union of Basic and Clinical Pharmacology (NC-IUPHAR) but also the citation of our most recent Concise Guide Introduction [2].
The collaboration between NC-IUPHAR and HGNC for standardising the nomenclature of pharmacological targets goes back well over a decade. This has been (and continues to be) an iterative process whereby NC (after extensive consultation) recommends naming schema for certain target classes that are different to those approved by HGNC. This is typically done on the basis of well established publication usage in the pharmacological community. In many cases these older protein family names predate completion of the human genome. This can be illustrated for the case of the Voltage-gated calcium channels as shown in the GtoPdb family page below.
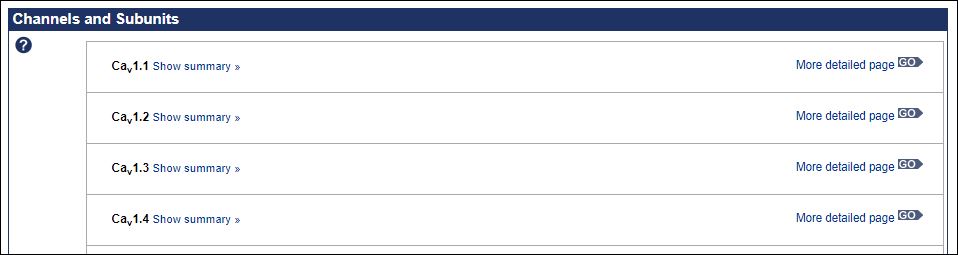
As we can see in the page for Cav1.1 curation adds a full set of synonyms including CACNA1S and “calcium voltage-gated channel subunit alpha1 S” as approved HGNC Symbol and name, respectively. Below we can see the reciprocal cross-pointing between HGNC and GtoPdb/NC-IUPHAR.


We have commented some time ago on ambiguity issues arising from the necessity of curatorial resolution for authors’ descriptions of key entities to standardized identifiers, including mapping between ligands and target protein names [3]. We consequently hope both papers will contribute to greater use of both HGNC and NC-IUPHAR nomenclature in the pharmacology literature.
Comments by Chris Southan, Fellow of the University of Edinburgh, Owner of TW2Informatics, Chair of NC-IUPHAR Subcommitees for Proteases and Drug Targets and Chemistry (DRUTACS).
[1] Kenji Fujiyoshi, Elspeth A. Bruford, Pawel Mroz, Cynthe L. Sims, Timothy J. O’Leary, Anthony W. I. Lo, Neng Chen, Nimesh R. Patel, Keyur Pravinchandra Patel, Barbara Seliger, Mingyang Song, Federico A. Monzon, Alexis B. Carter, Margaret L. Gulley, Susan M. Mockus, Thuy L. Phung, Harriet Feilotter, Heather E. Williams, and Shuji Ogino (2021) Opinion: Standardizing gene product nomenclature-a call to action, Proc Natl Acad Sci U S A Jan 19;118(3) doi: 10.1073/pnas.2025207118, PMID: 33408252.
[2] Stephen P H Alexander, Eamonn Kelly, Alistair Mathie, John A Peters, Emma L Veale, Jane F Armstrong, Elena Faccenda, Simon D Harding, Adam J Pawson, Joanna L Sharman, Christopher Southan, O Peter Buneman, John A Cidlowski, Arthur Christopoulos, Anthony P Davenport, Doriano Fabbro , Michael Spedding, Jörg Striessnig , Jamie A Davies, CGTP Collaborators (2019) The Concise Guide to Pharmacology 2019/20: Introduction and Other Protein Targets, Br J Pharmacol. Dec; 176 (Suppl 1): S1–S20, PMID: 31710719.
[3] Christopher Southan 1, Joanna L Sharman 1, Elena Faccenda 1, Adam J Pawson 1, Simon D Harding 1, Jamie A Davies 1 (2018) Challenges of Connecting Chemistry to Pharmacology: Perspectives from Curating the IUPHAR/BPS Guide to PHARMACOLOGY ACS Omega, Jul 31;3(7):8408-8420, PMID: 30087946.


Leave a comment